Summary of our research
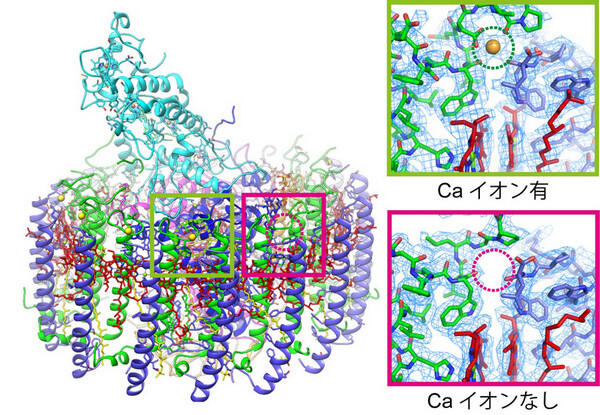
For many years we have been working on either Ca2+-bound or Ca2+-free light-harvesting (LH1) complexes from purple photosynthetic bacteria, with an overarching objective of revealing the minimum structural requirement for their highly selective Ca2+-binding. Following determinations of the structures for fully Ca2+-bound LH1 complexes, here we present a cryo-EM structure of a Ca2+-bound LH1 composed of multiple αβ-polypeptides in a single complex that, unlike our previous studies, originates from a species whose native LH1 contains a reduced number of Ca ions. Using this model, we have now been able to pinpoint the long-sought link between the two types of LH1 complexes and precisely define the minimum structural motif for specific Ca2+-binding; such binding is crucial for the biochemical and spectroscopic properties of the photocomplexes. This work is not "just another complex", but a complex from an unusual phototroph that has allowed us to answer an important and longstanding question that was not obvious from previous work. Our study lays the necessary foundation for advancing studies of selective metal-binding properties in other cellular systems and thus should appeal to a broad scientific audience, particularly those whose interests include metalloproteins, metal-related life science, bioenergetics, photosynthesis, structural biology, and the diversity of photosynthetic life on Earth.
Researcher information
TANI Kazutoshi
Appointed Professor, Mie University
Specialized area:
structural biology
MIZOGUCHI Akira
Professor, Mie University
Specialized area:
electron microscopy (Anatomy)
Zeng-Yu Wang-Otomo
Professor, Ibaraki University
Specialized area:
Photosynthesis
KIMURA Yukihiro
Associate Professor, Kobe University
Specialized area:
Photosynthesis