Summary of our research
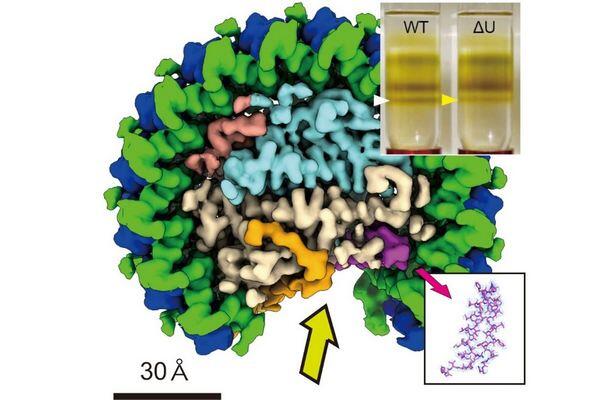
Rhodobacter sphaeroides is the "E. coli " of photosynthesis research because of the many fundamental insights it has provided to unraveling the biological mechanism of solar energy conversion. However, to our surprise, there are still important unknowns in the well-known. We show here that Rba. sphaeroides continues to reveal the most intimate new secrets of photosynthesis. Despite long-term efforts including our own work, a detailed structure of the core complex from this model system has not yet been achieved and is long awaited. Using cryo-EM, we have overcome previous roadblocks to create the first atomic-resolution picture of the Rba. sphaeroides LH1-RC complex. Our work provides a rare example that cryo-EM can directly identify an unreported protein in this complex that previously escaped from any biochemical analysis. The structure reveals for the first time the precise location and interactions of both the pigments and proteins that form the complex and addresses both structural and molecular aspects. Our study lays the necessary foundation for advancing studies of photosynthetic processes in Rba. sphaeroides and thus should appeal to a broad scientific audience, particularly those whose interests include photosynthesis and bioenergetics, structural biology, and the diversity of photosynthetic life on Earth.
Significance:
(1) Identified a previously unrecognized integral membrane protein and characterized its likely functions.
(2) Determined the precise position and conformation of the key protein PufX, settling a long-standing issue in bacterial photosynthesis.
(3) Resolved all carotenoid molecules in the core complex for the first time.
For more information, please click here.
Researcher information
TANI Kazutoshi
Appointed Professor: Mie University
Specialized area:
structural biology
MIZOGUCHI Akira
Professor: Mie University
Specialized area:
electron microscopy (Anatomy)
Zeng-Yu Wang-Otomo
Professor: Ibaraki University
Specialized area:
biological structural chemistry